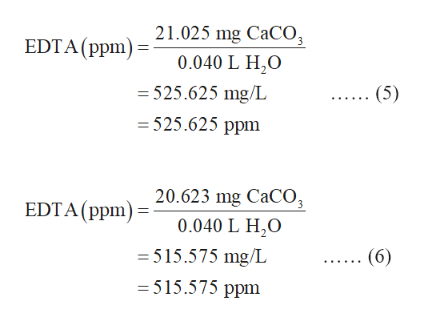
So, we can convert the volume of the solution to a mass:


Water can be assumed for your purposes to have a density of #"1 g/mL"#, and for this low of a concentration, we can assume that the density of water is equal to the density of the solution. Since we have a solution, let's suppose we are in a solution of water, which can dissolve small amounts of #"CaCO"_3# (normally "insoluble" in water, but at the ppm scale it is considered soluble). A #"ppm"# is a part-per-million, which is a concentration.
